Archive
Naltrexone for Cancer
Naltrexone is an opioid receptor antagonist approved and used for management of alcohol and opioid dependency. Low dose naltrexone or LDN (at 1/10th of the dose used for drug rehab) however has been proposed as an off-label therapy for a broad range of immune disorders such as HIV, MS, autoimmune thyroiditis, and colitis, and is one of the more popular off-label treatments for cancer thanks to some promising trials, low toxicity, relative low cost and much internet publicity such as found on the Low Dose Naltrexone Homepage and a Low Dose Naltrexone Forum. But if you don’t have a health science degree you might be wondering, what is the connection between cancer and opioids? And most importantly, does it work?
Opiates and Cancer
First some background on opioids and cancer. Firstly, there is the difference of endogenous opiates (eg endorphins and enkephalins) vs. exogenous opiates (drugs). The effects of opioids on cell growth is complex and is believed to be mediated through opioid and non-opioid receptor signalling (See Chen YL et al. The Other side of the Opioid Story: Modulation of Cell Growth and Survival Signalling, Curr Med Chem 2008:15(8):772-8), thus opioids directly modulate cell growth and endogenous opiates can directly suppress cancer growth. On the other hand, exogenous opiates can suppress the immune system, which is not ideal for treating cancer. It has been known for some time from animal studies that opioids interfere with the immune system ( Sacerdote P, Opioids and the Immune System, Palliat Med 2006;20 Suppl 1:s9-15), and that opioid pain killers such as morphine can decrease and depress immunity. In animal studies for example, morphine causes worsening of cancer, although the effect is different amongst different opioids, with buprenorphine (“Bupe”) perhaps the least immunosuppressive.
Background 1: Zagon
Now back to Naltrexone, an opioid antagonist. There is no doubt that Dr. Ian Zagon at Penn State is a major pioneer researcher in endogenous opiates and the major bench-side explorer of “off-label” applications of LDN. In his own words about the journey of discovery of opioid effects on cancer, he said : “When we discovered the effects of opioid antagonists such as naltrexone and naloxone in 1979, this was purely happenstance. Around 1975, we were interested in the effects of opiates… on children who were born to mothers that were addicted. The scientific literature revealed that these babies and children had neurological difficulties and were lower in body weight. We (myself and Dr. Patricia McLaughlin) developed a model to look at this in animals. Along the way, I was doing another project on neuroblastoma, a childhood tumor. When I found that these exogenous opioids altered growth of these developing animals… This started in 1977-1978. We then progressed to injecting cells into mice and creating cancers, and examined whether these exogenous opioids would repress growth of these cancers. In fact, they did…” (as quoted on the LDN forum).
It turns out from Zagon’s research, that the central actor may be one “OGF” or opioid growth factor, or otherwise known technically as [Met5]-enkephalin. Zagon proposed that OGF is an inhibitory peptide whose action is modulated via an OGF receptor and which modulates cancer cell proliferation and migration, and angiogenesis. Zagon has demonstrated that OGF inhibits pancreatic (BxPC-3), colon (HT-29), renal cell (Caki-2), neuroblastoma, and head and neck (CAL-27) cell lines (Int J Oncol, 2000 Nov;17(5):1053-61). Moreover, OGF also suppressed pancreas cancer in animals (Cancer Lett 1997 Jan 30;112(2):167-75) , and synergistically enhanced the efficacy of chemotherapy against pancreas cancer (Cancer Chemother Pharmacol 2005 Nov;56(5):510-20) and enhanced survival in squamous cell head and neck cancer as well(Cancer Chemother Pharmacol 2005 Jul;56(1):97-104). Based on such observations, Zagon & McLaughlin filed a patent in 1997 claiming that the administration of an opiate antagonist such as Naltrexone “at an amount sufficient to effect the intermittent blockade of the zeta receptor present in the cancer (and surrounding tissues) thereby producing a subsequent period of elevated endogenous enkephalin levels or receptor numbers to inhibit, arrest and even prevent tumor growth” (US Patent 6136780)
Background 2. Bihari
Almost working in parallel as Zagon, but from the clinical side and not in the laboratory, there is one Dr. Bernard Bihari, who is an addiction specialist who used Naltrexone and claims to have discovered the immunomodulatory benefits of Naltrexone in 1985. The story goes that Dr. Bihari began noticing in the 1980s that some of his addict patients with immune deficiency (subsequently discovered to be HIV/AIDS) had symptomatic improvement on lower doses of Naltrexone, so he conjectured that Naltrexone somehow upregulated their immune system (See AIDS Patient Care 1995 Feb;9(1):3). Along this line of thinking and based on some reports that lymphoma responded to endorphin treatment in animals, he had treated a recurrent lymphoma patient with low dose naltrexone and the lymphoma got better. He subsequently also treated a woman named CP with advanced melanoma and the cancer responded. Then from 1999 onwards, Dr. Bihari investigated the effects of LDN in private patients, using a low dose of 3mg given at night and theorizing that the treatment induced an increase in endorphins, especially metenkephalin, in the pre-dawn hours. The endorphins would in turn directly suppress cancer growth and upregulate the immune system. This theory coincided with Zagon’s animal work on how OGF may inhibit cancer and is consistent with the actions of naltrexone. Unfortunately, there has not been any organized clinical trial or even published case series on this, except for what has been presented in the Low Dose Naltrexone website, that “as of March 2004 … medication by Dr. Bihari in some 450 patients with cancer, almost all of whom had failed to respond to standard treatments, suggests that more than 60% of patients with cancer may significantly benefit from LDN. Of the 354 patients with whom Dr. Bihari had regular follow-up, 86 have shown objective signs of significant tumor shrinkage, at least a 75% reduction. 125 patients have stabilized and/or are moving toward remission” Apparently, of patients treated, “88 LDN-only group includes five breast cancer patients, one patient who had widespread metastatic renal cell carcinoma, three with Hodgkin’s disease and six with non-Hodgkin’s lymphoma. Reportedly, other cases, some on LDN for as long as four years, included a score of patients with non-small cell lung cancer, as well as patients with ovarian cancer, uterine cancer, pancreatic cancer (treated early), untreated prostate cancer, colon cancer, malignant melanoma, throat cancer, primary liver cancer, chronic lymphocytic leukemia, multiple myeloma and some others” according to another website. Again reportedly, in June 2002 an oncologist and an oncology physician’s assistant from the National Cancer Institute reviewed some 30 charts of cancer patients at Dr. Bihari’s office, and about half were chosen as appearing to have responded to LDN without question. Supposedly, copies of these records were sent to the NCI for further data collection on its part for consideration for NCI’s Best Case Series. Four cases of prostate cancers responding to LDN was reported as well in a patent “Method of Treating Cancer of the Prostate” Bihari filed in 2000. But again, regretfully, none of any of these cases ended up being published in the medical literature, and they circulate as quasi-anecdotal mentions online.
How might Naltrexone work in cancer?
In summary, based mainly on Zagon’s work, naltrexone at low dose administered nocturnally could bne postulated to work via 1) a stimulation of endogenous opiates as well as the number and density of opiate receptors on tumor cell membranes making them more responsive to the inhibitory effects of circulating opiates, which in turn suppresses tumor growth directly, 2) an enhancement of cellular immunity as a result of effects of higher levels of endogenous opiates, and 3) metabolites such as methylnaltrexone which exert antiangiogenic effects.
What does the medical literature say?
Bihari had published nothing on LDN and cancer in the medical literature.
Zagon had reported on using naltrexone in a mouse neuroblastoma model showing inhibition of growth and prolonged survival in those mice that develop tumors and protected some mice from developing tumors altogether (Brain Res 1989 Feb 20;480(1-2):16-28). At a similar dose of 0.1mg/kg, his team was also able to retard implanted human colon cancer in mice (Cancer Lett 1996 Mar 29;101(2):159-64), apparently via an stimulation of metenkephalins, which is in line of his research hypothesis.
Clinically, only two single case reports of 1) a long surviving metastatic pancreas cancer treated with LDN and alpha-lipoic acid (Integr Cancer Ther 2006; 5(1):83-9), and 2) a B-cell lymphoma with clinical reversal using only LDN Integr Cancer Ther 2007; 6(3):293-296) can be found in the medical literature that I can find.
Along these lines, an OGF plus gemcitabine for pancreas cancer trial is on the way (starting this month!) at Penn State, but while there are several trials on LDN for Crohn’s and MS and other conditions on the way, there is nothing on the horizon testing LDN for cancer per se.
My Take
I have been prescribing off-label Naltrexone to my cancer patients for many years. I remember being asked by organizers from Drs. Bihari’s camp to present at the 1st Annual LDN Conference in 2005 but declined to attend because I had no clear cut cases to report (Then they asked me to report on the tolerability of the treatment, which is not meaningful, and I didn’t go). Indeed, over the years, I have not seen any definitive responses that I can attribute to LDN with certainty. I wish to give some credence to the cases of response found online, but such are anecdotes that cannot strictly be considered admissible evidence in clinical science, only suggestive leads for further investigations. There are inherent limitations for testing LDN of course: not every patient is a candidate (eg if on narcotics for cancer pain) for LDN and most who take it are on at least a few other treatment modalities making a judgement of LDN efficacy difficult. Then also, the drug itself is cheap and generic and thus there is no industry interest in funding formal trials. However, I still prescribe LDN to this day since the theoretical background is not unsound, the side-effects are minimal, and the substance is readily available and at such a reasonably low cost that I usually do not mind the addition of LDN to a patient’s treatment, especially if requested and especially in cases of pancreas or colon ca, melanoma, SCCHN where there has been studies or prostate cancer where there has been a patent filed. Personally, I think LDN may perhaps have greater promise in other conditions such as Crohn’s and MS rather than cancer, and OGF may be a more direct opioid treatment option for cancer in the future.
Your comments welcome.
Gossypol (棉酚) for cancer
This is an unusual one because Gossypol it is not yet approved for us in the US or Europe. However, notwithstanding approvals or not, it is developed mainly as a male contraceptive. As such its potential application as a cancer therapy is vastly interesting and can loosely be classified as “off-label”.
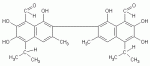
Gossypol is a polyphenolic compound isolated from the seeds, stems, and roots of the cotton plant (genus Gossypium, family Malvaceae; pls feel free to search under these in our Asian Anti-cancer Herbs database for more research data). It was discovered during the late 1960s, when people in rural China complained of fatigue attributable to exposure to cotton seed oil. Years later, many couples had fertility problems despite reduced exposure to the oil. The finding that exposure to cotton seed oil was related to lowered sperm counts in men exposed led to the hypothesis its active ingredient Gossypol could be used as a male fertility-control agent, which is gossypol’s main pharmacologic application now. Of course, a natural line of thinking with an agent that may inhibit dividing or growing cells such as sperm would lead one to query if it may have chemotherapeutic properties that may be applicable in cancer, so this is where the idea of Gossypol for cancer arises.
And where is the evidence?
a) In Vitro (cellular evidence):
Russian scientist Vermel EM et al. reported on the anti-cancer activity of gossypol in animals as early as 1963 and Jolad SD et al reported in 1975 (J Pharm Sci 64:11, pp.1889-1890, 1975) that Gossypol extracted from Montezuma speciosissima Sesse and Moc. demonstrated tumor-inhibiting properties in the P-338 lympocytic leukemia test system (3PS).
Gossypol promotes apoptosis of breast, bladder, lymphoma, leukemia (CML and CLL), myeloma, prostate, colorectal, alveolar cell lung, glioma, pancreas, melanoma, nasopharyngeal, and head and neck squamous cell cancers. A preponderance of the research reported on efficacy against hematologic cancers and prostate cancer.
Gossypol was known in the 1990s as a compound which depleted cellular energy by inhibition of intracellular dehydrogenases. More recently, (-)-Gossypol, now understood to be a natural BH3 mimetic, is found to be a small-molecule inhibitor of Bcl-2/Bcl-xL/Mcl-1, possibly exerts its antitumor activity through inhibition of the antiapoptotic protein Bcl-xL accompanied by an increase of proapoptotic Noxa and Puma (Meng Y, et al. Mol Cancer Ther, 7:7, pp. 2192-2202, 2008). A separate line of evidence suggests that Gossypol may exert its apoptotic effects via downregulated expression of NF-kappaB-regulated gene products, including inhibitor of apoptosis protein IAP-1, IAP-2, and X-linked IAP (Moon DO, et al. Cancer Lett 264:2, pp.192-200, 2008).
It is also known as a protein kinase C inhibitor.
Besides direct anticancer action, it enhances anti-tumor activity of chemotherapy against lymphoma, modulates multi-drug resistance gene expression in human breast cancer cells, and enhanced breast cancer sensitivity to Tamoxifen as well as Adriamycin.
In Vivo (animal evidence):
Testing of gossypol on tumor growth and the survival of 10- to 12-week-old BDF1 mice bearing injected mammary adenocarcinoma 755 (Ca 755) or P388 or L1210 leukemias was investigated and reported as early as 1985.
Gossypol enhances prostate cancer response to radiation therapy (mice), enhances chemotherapy against diffuse large cell lymphoma in WSU-DLCL2-SCID mouse model pre-clinical testing.
Many other reports of in vivo activity of Gossypol against transplanted tumors in rodents exists.
Clinical (human evidence):
Much clinical experience of Gossypol’s anticancer use and demonstration of its efficacy has been accumulating in the past 20 years.
One of the earliest trials was reported from the U.K. (Stein RC, et al. Cancer Chemother Pharmacol 30:6, pp. 480-2, 1992) where advanced cancer patients were given Gossypol, but benefit was not seen.
In 1993, the NIH published a trial of oral Gossypol using doses of 30-70 mg a day in metastatic adrenal cancer patients where 30% of eligible patients had some reponse to therapy (Flack MR et al., J Clin Endocrinol Metab 76:4, pp. 1019-24, 1993). A study was also carried out using low dose Gossypol of 10mg twice a day on adults with heavily pre-treated, poor prognosis recurrent malignant gliomas and found approximately 25% with some response including one patient who remained stable with improved quality of life for one and a half years. More importantly, toxicity was found to be mild.
Around the same time, a Phase I/II clinical trial of Gossypol against refractory metastatic breast cancer was carried out at Memorial Sloan-Kettering in New York ( Van Poznak C, et al. Breast Cancer Res Treat, 66:3, pp. 239-248). Doses were in the 30-50mg per day range with 30% of patients experiencing fatigue, 15%, nausea/vomitting, and diarrhea in 10%. Antitumor activity was seen with a 15% response/stability rate.
Significantly, long-term clinical remission of a patient with chronic lymphocytic leukemia using gossypol was reported. (Politzer, Phytomedicine 15:8, pp. 563-5, 2008)
So What’s new?
Gossypol is currently the only orally bioavailable pan-Bcl-2 inhibitor under clinical investigation.
In the US, phase I/II clinical trials are currently ongoing or planned with gossypol under the product code “AT-101” by Ascenta Therapeutics as an adjuvant therapy for human prostate cancer. A small trial of the product in 23 men with prostate cancer who were chemo-naive but had rising PSA and who took 30mg of product for 21 out of 28 days over 20 to 24 weeks noted decreases in PSA parameters in some. 5 out of the 23 patients had to discontinue the drug because of gastrointestinal side-effects (ileus). Last summer, Ascenta also presented preliminary data indicating that AT-101 has activity in combination with taxol and prednisone in advanced prostate cancer.
The biotech firm Bioenvision (now owned by Genzyme) was in collaboration with Bowman Research to develop a process to separate and purify more efficacious but less toxic iso-forms of gossypol, but the isoforms are not currently in the company’s development pipeline.
My Take
This is a fairly low toxicity and inexpensive natural derivative that can be used in combination with chemotherapy or other anti-apoptotic agents for a wide-range of cancers but appear especially promising for prostate, breast and B-cell hematologic malignancies. The main concern here is side-effects which include fatigue, nausea/vomitting, diarrhea, and ileus. Side-effects can be managed by individualized dosing and schedule adjustments. The long-term concern of infertility in males should be considered in young male patients. The lack of bone marrow suppression makes it a good agent to combine with with chemotherapies. The potential usefulness of an agent like this points the way to screen other anti-fertility herbs in traditional pharmacopoeias for other potentially useful anti-cancers. Additionally, this interesting natural derivative has been demonstrating promise as an anti-HIV, as well as anti-malarial . (More on the very interesting off-label potential of a whole range of anti-malarials against cancer in this blog in the near future, when I get to it) . Your comments please !
Recent Comments